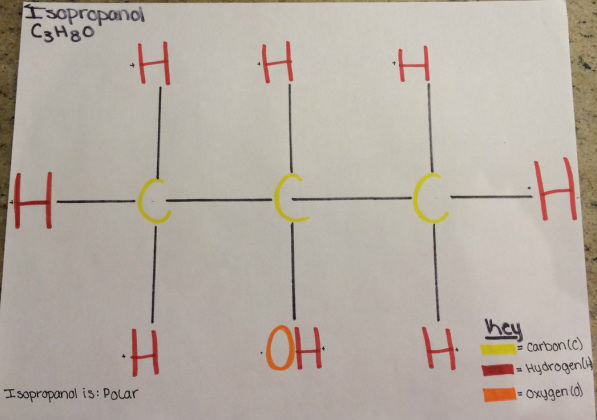
Drawing of Isopropanol
Polarity
Isopropanol, C3 H8O is a polar molecule. This is because all of the hydrogen atoms have a positive charge but because there is one oxygen bonded to a hydrogen that the oxygen has electrons open making it have a negative charge. Therefore isopropanol is polar because of the uneven distribution of positive and negative charges.
Isopropanol, C3 H8O is a polar molecule. This is because all of the hydrogen atoms have a positive charge but because there is one oxygen bonded to a hydrogen that the oxygen has electrons open making it have a negative charge. Therefore isopropanol is polar because of the uneven distribution of positive and negative charges.